Third International Scientific and Practical Conference “Eagles of the Palearctic: Study and Conservation”
Raptors Conservation. Suppl. 2. Proceedings of Conferences
VETERINARY DRUGS AND ASSOCIATED IMPACT ON VULTURE HEALTH IN ASIA
Bohra D.L. (Department of Zoology, Seth G.B. Podar College, Nawalgarh, Jhunjhunu, Rajasthan, India)
Contact:
Dau Lal Bohra daulalbohara@yahoo.com
Recommended citation: Bohra D.L. Availability of Veterinary Drugs Residues and Associated Impact on Vulture Health in Asia. – Raptors Conservation. 2023. S2: 175–177. DOI: 10.19074/1814-8654-2023-2-175-177 URL: http://rrrcn.ru/en/archives/35001
There are over 500 million livestock in India, and without infrastructure to dispose of dead livestock animals, pastoralism has historically depended on vultures (Gyps sp.) as environmental sanitizers. In their absence, carcasses of dead animals spend time out in the open or are disposed by farmers in water, both of which create an increased risk of disease and water pollution. This carrion also creates a new source of food for dogs and rats, increasing their population. Feral dogs and rats are a major source of rabies infections a known public health issue in India. Rabies infection is lethal unless vaccination immediately after exposure. The collapse in India’s vulture population occurred because of unintended poisoning following the availability of cheap generic versions of diclofenac for human use till 2008.
Diclofenac was introduced in 1973 as a painkiller for humans, but in the mid-90s, along with approval for a generic version in 1993, the pharmaceutical industry in India started producing large quantities of the drug. This lowered the price to a point that made diclofenac use in livestock economically viable and by 1994, diclofenac was widely available across veterinary clinics. Diclofenac was the first NSAID shown to be toxic to scavenging birds and has been banned for veterinary use in much of Asia vultures’ range. Currently, there are fully gazetted bans on the manufacture, sale, and use of veterinary diclofenac in Bangladesh, Cambodia, India, Iran, Nepal, Oman, and Pakistan, and other countries are considering a similar ban. With the intervention of Human right commission in India, generic versions of multi dose packs of diclofenac for human use was banned in 2015 to stop misuse in animals treatments.
In 2023, Ketoprofen, Aceclofenac more fully gazetted bans on the manufacture, sale and use of veterinary for vulture protection. Other then Diclofenac, Ketoprofen, Aceclofenac, two more agents including Nimesulide and their composition, and Flunixin are toxic to vultures. As per scientific reports and analysis in safety testing experiments have established that meloxicam and tolfenamic acid are safe. Use of medicines for One Health issue, and the use of veterinary medicines can have consequences affecting animal health, welfare, and ecosystems. According to Nambirajan, 2018 range of diclofenac 62.28 to 272.20 ng/g in 32 dead White-Backed Vultures (Gyps africanus). In another similar incidence, 14 White-Backed Vultures had diclofenac in kidneys in toxic range (70–908 ng/g), and in 12 Himalayan Griffons (Gyps himalayensis), diclofenac in the range of 139.69 to 411.73 ng/g.
In 2021, a new drug has caused four White-Rumped Vulture (Gyps bengalensis) deaths as nimesulide was detected in all the tissues (17–1395 ng/g). As veterinary aspects are critical to stakeholders of pharmaceutical industry, and we can leverage our multiple spheres of influence to help mitigate the animal and public health, as well as reduce the ecological footprints of medicine use. In addition, there is also a need to analyze the drug influence on vulture reproductive health in Central Asia.
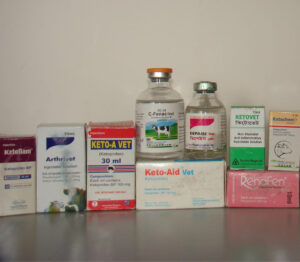
Veterinary non-steroid anti-inflammatory drugs, which have caused a catastrophic decline in vulture populations due to poisoning, sold in veterinary pharmacies in India. Photo by D. Bohra.